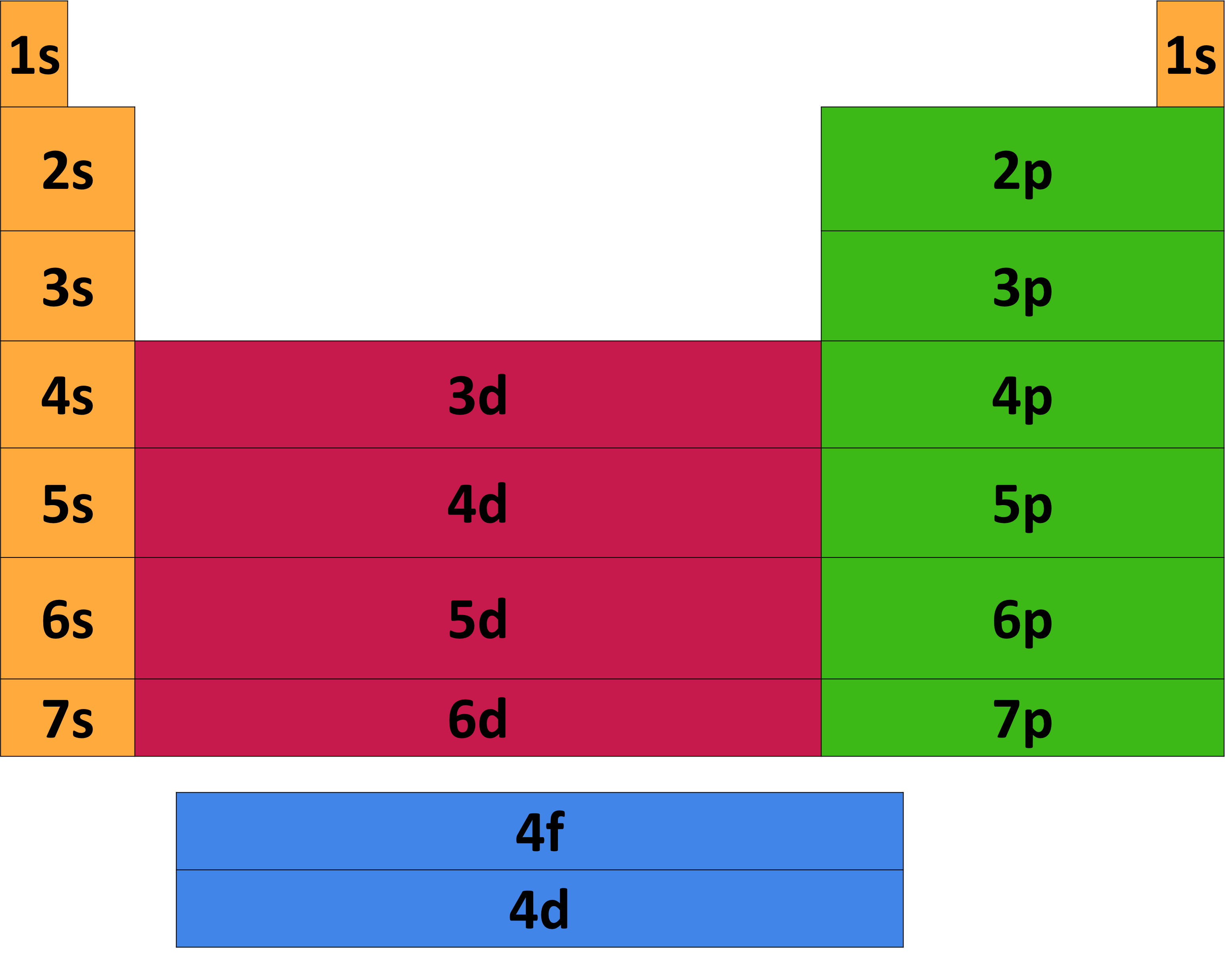
Electron configurations and orbital diagrams can be determined by applying the Pauli exclusion principle (no two electrons can have the same set of four quantum numbers) and Hund’s rule (whenever possible, electrons retain unpaired spins in degenerate orbitals).Įlectrons in the outermost orbitals, called valence electrons, are responsible for most of the chemical behavior of elements. Atomic radius defined as half the distance between adjacent nuclei in a crystal of an element Electronegativity indicates the relative ability of its atoms to attract electrons in a chemical bond.The relative energy of the subshells determine the order in which atomic orbitals are filled (1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, and so on). Gases most can conduct electricity.remember Cathode-Ray tubes and neon signs First Ionization energy energy required to remove the first electron from an atom. They take the shape volume of their container. Gases floating elements that have no definite shape or volume. Non-metals are mostly powders that are non-conductors of electricity. Metalloids located between the metals and the gases along the zigzag line of "p" block. Metalloids are like metals as some are able to conduct some electricity (a.k.a. Metals make up the majority of periodic table Metals defined as good conductors of electricity, have higher luster (shine) and are malleable (capable of being beat, using hammer or rollers, into wires). Octet full valence shell or 8 electrons in a shell. Noble gases the family in column 18 Noble gases the least reactive elements on the table, as they possess a full valence shell known as an octet. Main-group elements The "s" and "p" blocks collectively together are known Halogen family family in column 17 Halogen family are very reactive elements as they are missing 1 electron from having a full valence shell. Metalloids are next to the zigzag line running down through the "p" block in the table. Transitional Metals elements Families 3-12 Transitional Metals (families 3-12) make up the "d" block/orbital "P" block elements Families 13-18 "P" block elements a collection of metals, metalloids, non-metals, and gases. 
This is bc family 1 has 1 valence electron and family 2 has 2 valence electrons. Alkali Metals family Family 1 Alkaline Earth metals family Family 2 Families 1 and 2 are in the "s" block/orbital Families 1 and 2 are highly reactive with air, moisture, or most other elements. 18 # families on periodic table Family have very similar chemical properties. radioactive gives off energy Families Vertical columns of table. Synthetic Elements man-made in the laboratory and radioactive. Actinide series portion of the "f" block in the seventh period. Lanthanide series portion of the "f" block in the sixth period. Sixth period has "s", "f", "d", p" orbitals. "p" Block has 3 sub-orbitals "d" Block has 5 sub-orbitals Fifth period has the same as the fourth.has "s", "d", and "p" orbitals. Each "P" block has 3 sub-orbitals fourth period has an "s", "d" and "p" orbitals. Each "P has 3 sub-orbitals Orbitals can only hold 2 electrons third period just like second.has "s" and "p" orbitals. Periodic Table appears as Rows and Columns Rows called periods First period only has "s" orbital second period has an "s" and "p" orbitals. Nole Gases the least reactive group of elements on the Periodic Table Noble Gases have a full valence (outer most) electron energy shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
